Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Lee, C. G.; Suzuki, Daisuke; Kokubu, Yoko; Esaka, Fumitaka; Magara, Masaaki; Kimura, Takaumi
International Journal of Mass Spectrometry, 314, p.57 - 62, 2012/03
Times Cited Count:22 Percentile:75.59(Physics, Atomic, Molecular & Chemical)In this study, we developed a method for the simultaneous measurement of all the plutonium and uranium isotopes without the need for chemical separation, using thermal ionization mass spectrometry (TIMS) with a continuous heating method. The MOX particles with sizes of 0.6-2.3  m used in this study were made from a mixed solution of plutonium (SRM947) and uranium (U500). The isotope ratios of plutonium and uranium obtained for all the MOX particles, including the ones in the sub-micrometer size range, were in good agreement with the certified values within the range of error. In particular, the determination of the
m used in this study were made from a mixed solution of plutonium (SRM947) and uranium (U500). The isotope ratios of plutonium and uranium obtained for all the MOX particles, including the ones in the sub-micrometer size range, were in good agreement with the certified values within the range of error. In particular, the determination of the  Pu/
Pu/ Pu isotope ratio, which is difficult because of the isobaric interference of
Pu isotope ratio, which is difficult because of the isobaric interference of  U, was performed accurately. The
U, was performed accurately. The  Pu/
Pu/ Pu ratios were obtained by using a correction in which the background of
Pu ratios were obtained by using a correction in which the background of  U intensity at the
U intensity at the  Pu peak was estimated from the peak fitting of the
Pu peak was estimated from the peak fitting of the  U signal profile.
U signal profile.
Kokubu, Yoko; Suzuki, Daisuke; Lee, C. G.; Inagawa, Jun; Magara, Masaaki; Kimura, Takaumi
International Journal of Mass Spectrometry, 310, p.52 - 56, 2012/01
Times Cited Count:20 Percentile:72.67(Physics, Atomic, Molecular & Chemical)A method for measuring isotope ratios of Pu and U in Pu or Pu/U mixture samples was developed by using a continuous heating method of thermal ionization mass spectrometry (TIMS). In this method, temperature of an evaporation filament is increased sequentially. This leads to measure the ratios of Pu and U at different temperature in which each element is evaporated efficiently. In Pu/U mixture samples with pg-level masses and 0.01-10 Pu/U ratios, isotope ratios of 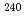 Pu/
Pu/ Pu and
Pu and  U/
U/ U were measured simultaneously without chemical separation of samples. The relative standard deviation of the isotope ratios of
U were measured simultaneously without chemical separation of samples. The relative standard deviation of the isotope ratios of 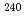 Pu/
Pu/ Pu and
Pu and  U/
U/ U in the sample containing each 1 pg of Pu and U were better than 2% and 4%, respectively. Moreover,
U in the sample containing each 1 pg of Pu and U were better than 2% and 4%, respectively. Moreover,  Pu/
Pu/ Pu ratios were obtained. The results suggest that the continuous heating method of TIMS will be effective tool as a method for measuring isotope ratios in the Pu/U mixture samples.
Pu ratios were obtained. The results suggest that the continuous heating method of TIMS will be effective tool as a method for measuring isotope ratios in the Pu/U mixture samples.
Suzuki, Daisuke; Kokubu, Yoko; Sakurai, Satoshi; Lee, C. G.; Magara, Masaaki; Iguchi, Kazunari*; Kimura, Takaumi
International Journal of Mass Spectrometry, 294(1), p.23 - 27, 2010/06
Times Cited Count:40 Percentile:87.1(Physics, Atomic, Molecular & Chemical)A new method for isotope ratio measurement with thermal ionization mass spectrometry (TIMS), "continuous heating method", was developed in order to determine an accurate isotope ratio of uranium ranging from sub-picograms to several dozen picograms. In this method, signals were measured during evaporation of an entire sample; however, part of the higher detected signals was used for calculation of the isotope ratio. The continuous heating method can measure the ratios with high reproducibility regardless of the sample amount and the skill of operators because procedures of measurement and data calculation were standardized, enabling accurate correction of mass discrimination effect. When  U/
U/ U ratios in isotopic reference materials measured by the continuous heating method were compared with those measured by the conventional method and the total evaporation method, there was no difference in the case of measurement of a larger amount of samples. However, the continuous heating method showed the most accurate result compared with those measured by the other methods for the measurement of the sample of sub-picograms.
U ratios in isotopic reference materials measured by the continuous heating method were compared with those measured by the conventional method and the total evaporation method, there was no difference in the case of measurement of a larger amount of samples. However, the continuous heating method showed the most accurate result compared with those measured by the other methods for the measurement of the sample of sub-picograms.
Hayakawa, Shigeo*; Harada, Kengo*; Watanabe, Nobuaki*; Arakawa, Kazuo; Morishita, Norio
International Journal of Mass Spectrometry, 202(1-3), p.A1 - A7, 2000/10
Times Cited Count:16 Percentile:60.07(Physics, Atomic, Molecular & Chemical)no abstracts in English